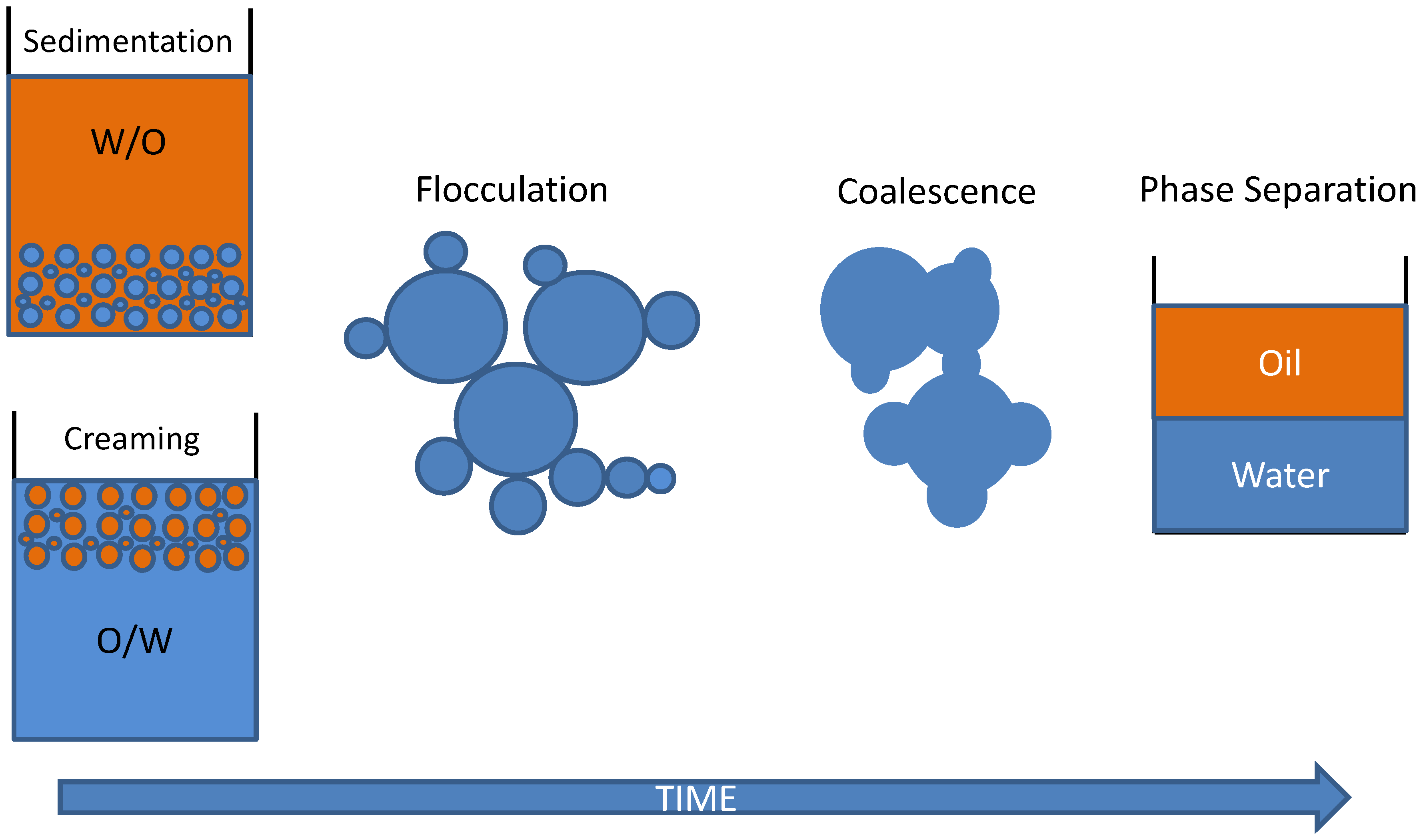 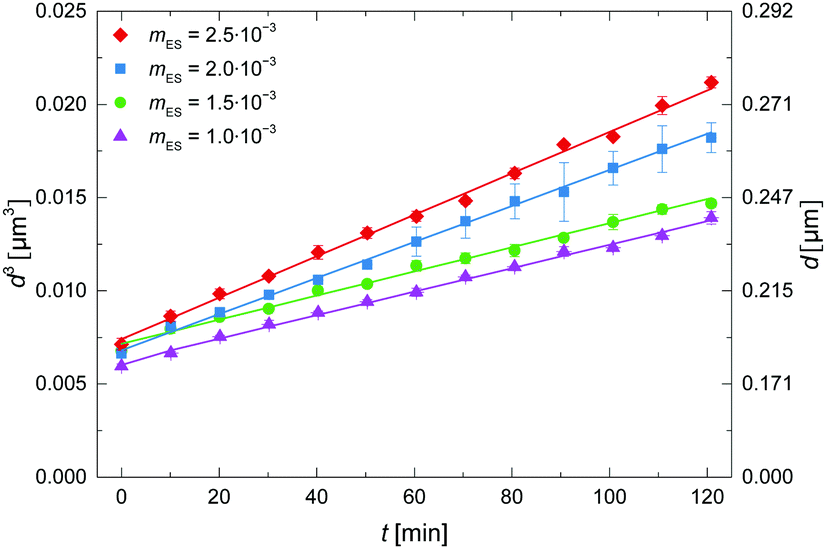
HLB values for commercial emulsifying agents range from 1 to 20. The emulsifying capability of an agent can be assessed through the hydrophile-lipophile balance (HLB), which is defined as the ratio of the weight percentage of hydrophilic groups to the weight percentage of hydrophobic groups in the molecule. Meanwhile, the ionic ones act through electrostatic repulsions, preventing droplets from approaching each other. Neutral surfactants mainly create steric barriers that prevent emulsion droplets from breaking down. Įmulsions are thermodynamically unstable systems, but they can be stabilized kinetically for some time (minutes to years) by adding surfactants that are adsorbed at the oil-water interface, creating a narrow (2–20 nm thick) three-dimensional layer surrounding the emulsion droplets and causing repulsive forces that provide some kinetic stability. Table 1 shows examples of typical food emulsions. Butter, margarines, and fat-based spreads are typical examples of w/o emulsions, and their properties depend largely on the properties and particular composition of the fats or oils and the surfactant used in the water phase. They are present in many dairy foods, including mayonnaise, creamers, whippable toppings, ice creams, etc. The o/w emulsions are probably the most versatile, and their properties are controlled by both the surfactants and the composition of the aqueous phase. Other emulsions that are employed include water-in-oil emulsions (w/o), where water droplets are dispersed in an oil, and water-in-oil-in-water (w/o/w) emulsions, which consist of an o/w emulsion whose droplets themselves contain water droplets. Anionic surfactants such as sodium dodecyl sulphate (SDS) are less frequently used to avoid electrostatic repulsions with the electrolytes present in water (e.g., NaCl, H 3O +), and cationic surfactants such as cetyltrimethylammoium bromide (CTAB) are never used.Įmulsions are widely used in the preparation of emulsified foods because lipids are mostly present in foods in the form of oil-in-water emulsions, where the oil droplets are dispersed in an aqueous solution. For this purpose, a recently developed methodology was employed, and experimental results were interpreted on the grounds of a pseudophase kinetic model.Ĭhemical structures of nonionic and zwitterionic surfactants commonly employed in the preparation of foodstuffs. In this work, we analyze the effects of the surfactant Tween 20 on the oxidative stability and on the effective concentrations of two model antioxidants derived from cinnamic acid, determining their interfacial concentrations in the intact emulsions to avoid disrupting the existing equilibria and biasing results. The rate of the inhibition reaction depends on the effective concentrations of antioxidants, which are mostly controlled by the amount of surfactant employed in the preparation of the emulsion. This is the case, for instance, in the inhibition reaction between antioxidants and the lipid radicals formed in the course of the spontaneous oxidation reaction of unsaturated lipids, which are commonly employed in the preparation of food-grade emulsions. However, addition of surfactants to binary oil-water mixtures also brings up the formation of three-dimensional interfacial layers, surrounding each emulsion droplet, that significantly alter chemical reactivity. They play two main roles in the emulsification processes: first they decrease the interfacial tension between the oil and water, facilitating droplet deformation and rupture second, they reduce droplet coalescence by forming steric barriers. Surfactants have been used for decades in the food industry for the preparation of lipid-based emulsified food stuffs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
